TOOKAD Vascular Targeted Photodynamic Therapy for Prostate Cancer at Vivantes Clinic Am Urban
Berlin, Germany


Overview
Age group
Adults
Type of care
Inpatient, Outpatient
Method
Non-surgical
About the offer
What’s included
Medical service
Examination
- clinical history-taking
- medical records review
- physical examination
- consultation with a urologist-oncologist
- consultation with anesthesiologist
Laboratory tests
- complete blood count (CBC)
- biochemical analysis of blood (kidney and liver function tests, electrolytes)
- inflammation blood tests
- PSA
- urinalysis
Diagnosis
- multiparametric MRI (mpMRI) of the prostate gland
- fusion-guided biopsy analysis (on the indication)
Treatment
- pre-procedure patient preparation
- personalized 3D dosimetric treatment planning using TOOGUIDE software
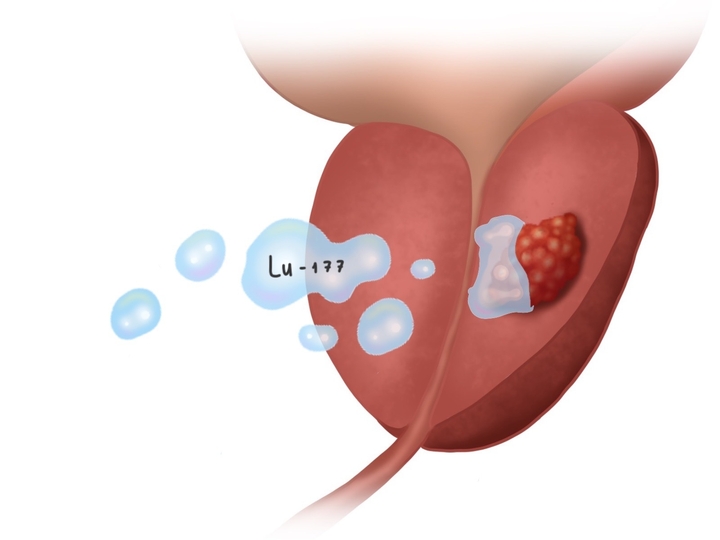
- TOOKAD VTP procedure (near-infrared laser application via transperineal optical fibers) under intra-operative ultrasound guidance and precision monitoring
- short-term light protection protocol
- post-procedure care
- symptomatic treatment
Other
- doctor's fee
- cost of essential medicines
- cost of essential materials
- hospitalization (up to 3 days)
- nursing service
- discharge medical records
- further recommendations
Extra add-ons
Airport transfer
Single room
Hospital meals
Personal coordinator
HealthDocs translate
AiroCare
AiroMedical support
Payment protection
Patient advocacy
Price match
Meet the provider
Gallery
Customize this offer
If you're not seeing exactly what you need here, send your custom request. You can discuss the content, specifics, price & timeline to create a personalized plan.
Location
Dieffenbachstraße 1, 10967 Berlin, Germany
FAQ
What is the rating of the offer?
TOOKAD Vascular Targeted Photodynamic Therapy for Prostate Cancer at Vivantes Clinic Am Urban is rated as 9.50 by AiroMedical.
Who is offering a deal?
The provider is Vivantes Clinic Am Urban.
How long does the offer take?
The offer is designed for 5 days.
Individual cost estimate. Non-binding 100% free assessment.
HomeOffersTOOKAD Vascular Targeted Photodynamic Therapy for Prostate Cancer at Vivantes Clinic Am Urban